UCB
-
In Same Day, UCB Lands Two FDA Drug Approvals in Autoimmune Diseases
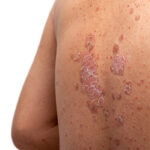
FDA approval of UCB’s Bimzelx gives the Belgian drugmaker a contender in the crowded plaque psoriasis therapies market. In the rare disease generalized myasthenia gravis, the agency approved Zilbrysq, a UCB drug with advantages over two blockbuster AstraZeneca drugs addressing the same target.
-
UCB Muscle Disease Drug Wins FDA Nod on Heels of Rival’s Regulatory Approval
The FDA approved UCB’s Rystiggo for treating generalized myasthenia gravis. Drugs are already available for the rare neuromuscular disorder, but Rystiggo’s approval includes treatment of a subset of patients not addressed by other products.
-
Jorie Healthcare CEO Shares Why Automation is Critical to Revenue Cycle Management
The revenue cycle management business is using AI tools to automate cumbersome tasks to help hospitals operate more efficiently. It’s beginning to attract the attention of major healthcare organizations.
-
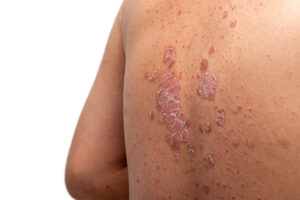
UCB’s FDA setback delays competition in U.S. with blockbuster J&J psoriasis drug
The FDA cited manufacturing issues in turning down UCB’s application seeking approval for bimekizumab, an antibody drug developed to treat moderate-to-severe plaque psoriasis. Bimekizumab is already approved and available in other markets around world, but the FDA rejection delays UCB’s antibody drug from competing in the U.S. for now.
-
AbbVie makes neuro drug connection with $130M Syndesi Therapeutics acquisition
AbbVie has acquired Syndesi Therapeutics, an early clinical neuroscience startup developing drugs that have potential applications in a range of cognitive disorders. Syndesi is based on research from Belgian pharmaceutical company UCB.
-
UCB is expanding its reach in epilepsy with $1.9B Zogenix acquisition
Epilepsy is a small part of UCB’s neurology pipeline, but its proposed Zogenix acquisition would broaden the Belgium-based drugmaker’s ability to address that market. Zogenix’s Fintepla is approved in the U.S. and Europe for a rare type of epilepsy called Dravet syndrome, and is in development for other rare epilepsies.
-
Novartis boosts neuro pipeline through alliance on UCB Parkinson’s drugs
Novartis is paying UCB $150 million up front to share in the development of two clinical-stage Parkinson’s disease drugs that offer new approaches to treating the disease. Depending on the progress of those drugs, UCB could earn up to $1.5 billion in milestone payments.
-
Artificial Intelligence, BioPharma
Belgium’s UCB to incorporate Accenture’s AI tech into pharmacovigilance efforts
The Belgian drugmaker will use Accenture’s INTIENT Pharmacovigilance to monitor the safety of its drugs, including those that are on the market and in clinical trials. Germany’s Bayer inked a similar deal with Genpact last year.
-
UCB psoriasis drug beats J&J’s second top-selling product in Phase III study
Results of the BE VIVID study showed that UCB’s bimekizumab showed a statistically significant improvement in skin clearance compared with J&J’s Stelara and placebo.
-
UCB to acquire Ra Pharmaceuticals for $2.1B
The Brussels-based drugmaker said the acquisition of Ra would boost its potential for leadership in the autoimmune disease myasthenia gravis, as both companies have drugs in Phase III development for it.
-
Navigating Healthcare’s Data Revolution: Priorities, Opportunities, and Challenges for Health Systems
Arcadia recently partnered with HIMSS Market Insights to survey executives, IT, technology, and clinical leaders. Here’s what we found.
-
Healthcare analytics firm Aetion gets $27M from biopharma, healthcare companies
The company plans to the use the money to extend its reach into therapeutic area-related intelligence and outcomes-based contracting for payers.
-
Patient engagement reaches the C-suite
In less than a decade, the chief patient officer – and similar roles with different titles – has become a key part of the healthcare industry executive team.
-
UCB’s off-label marketing of epilepsy drug Keppra costs $34M fine (Morning Read)
Current medical news from today, including: UCB reaches settlement over off-label marketing of its epilepsy drug Keppra; the Food and Drug Administration warns of higher prostate cancer risks from baldness and prostate drugs; flu shots reach an all time high; and UCLA partners with Roche for stem cell and cancer research.
-
Generic of UCB allergy med Xyzal gets preliminary FDA approval for Synthon
Generic drug company Synthon Pharmaceuticals has received tentative U.S. Food and Drug Administration approval on […]